+91-9240214610 (Bandra)
+91-9240214611 (Kandivali)





August 19, 2019 | Author: Admin
Purpose: To assess the effectiveness and tolerability of GenTeal® gel in post-menopausal patients with moderate to severe dry eye. Methods: This was an open label, multicenter, non-comparative, non-interventional, observational study in post-menopausal women patients of age 39-82 years with moderate to severe dry eye. The patients were treated with GenTeal® gel for 20 weeks and assessed for effectiveness at baseline, 10-12 weeks and at 20 weeks. The primary effectiveness outcomes were changes in ocular symptoms (foreign body sensation, itching, burning, watering, photophobia, feeling of dryness in the eye), tear break up time (TBUT), Schirmer, fluoresce in corneal staining score and global assessment for efficacy on dry eye condition. The secondary objective of the study was to evaluate ocular tolerance and systemic safety of the product.
Results: A total of 169 out of 170 enrolled patients completed the study. At 20 weeks of treatment, the median composite ocular symptoms scores was reduced by 78% from baseline (median; 2.0 vs 9.0 of baseline, p<0.0001). All ocular symptoms except photophobia were significantly reduced (p<0.05) at 20 weeks of treatment. At 20 weeks, TBUT and schirmer scores were significantly increased by 39.9% and 48.6% respectively (p < 0.0001); while fluoresce in staining cumulative score was significantly decreased (100.0%, p < 0.0001) from their baseline. The overall efficacy and ocular tolerability of GenTeal® gel, was ‘good’ to ‘excellent’ for >98.0% of the subjects. Adverse events of mild dyspepsia and frequent micturition of moderate intensity were reported by a subject. Conclusions: The findings of the present study indicate that GenTeal® gel treatment is an effective and tolerable treatment for dry eye in post-menopausal women patients in Indian clinical practice.
Dry eye is a multifactorial disease of the tears and the ocular surface, and recognized as a growing public health problem1-4. According to the report of the Epidemiology Subcommittee of the 2007 Dry Eye Work Shop, 14% -33% of the population worldwide suffers from dry eye5. The prevalence of dry eye disease for people e” 50 years is more among women (7.8%) than in men (4.7%) in the US2, 6. Dry eye is caused by a deficiency in the quantity and/or quality of tears, an unstable tear film and ocular surface damage leading to symptoms such as ocular irritation, dryness, fatigue, and fluctuating visual disturbances7.
Among the various risk factors associated with dry eye increasing age, female gender, androgen deficiency and hormone replacement therapy4 are vital for development of dry eye disease8. In post-menopausal women, conjunctival epithelium morphology is modified by estrogen deficiency, in particular by reducing the density of goblet cells responsible for mucous secretion, leading to instability of the tear film and dryness9.
The Currently recommended therapies for dry eye disease are lifestyle changes and the use of artificial tears products, anti-inflammatory medications or surgery10-13. These therapies however, alleviate symptoms but provides only temporary relief and blurring of vision leading to poor compliance14. The therapeutic goal is to control of the disease process in order to preserve vision and to provide comfort15.
GenTeal® gel (0.3% HPMC, 0.22% carbomer gel) is long-acting eye gel that has been approved by the US FDA for treatment of moderate to severe dry eye disease16. GenTeal® gel has disappearing preservative, sodium perborate which breaks down into water and oxygen upon contact with the tear film and ocular tissue thus lets patients with preservative sensitivities.
Previously, clinical trials have demonstrated the efficacy and tolerability of GenTeal® gel for treatment of dry eye disease in the United States17-18. It lubricates the surface of the eye with minimal blurring of vision as well as provides superior relief from the signs and symptoms of dry eye as in contrast to aqueous artificial tear substitutes. Additional evidence is needed to verify the effectiveness and tolerability of GenTeal® gel in moderate to severe dry eye disease among post-menopausal women. Thus the present open label, non comparative study was designed to assess the efficacy and tolerability of GenTeal® gel in post-menopausal women with moderate to severe dry eye disease prescribed during routine clinical practice in India.
Study design and patients
This was an open label, multi-center, non-comparative, non-interventional, 20 weeks observational study in India.
The study was performed in 170 post-menopausal women of age between 39-82 years with moderate to severe dry eye. Post- menopause was defined as absence of menstrual period for at least one year, or surgical hysterectomy with bilateral oophorectomy, no less than six months prior to the study. The subjects enrolled were either drug naïve patients or those who were previously treated with or on any other tear substitute when in the ophthalmologist’s opinion, they would benefit from GenTeal® Gel. The study was conducted according to the ethical principles of the Declaration of Helsinki and was approved by the Independent Ethics Committee or Institutional Review Board for each center. A written informed consent was obtained from all subjects prior to participation in the study.
Patients, who were on any other tear substitute, the usages of existing tear substitute was stopped, and switched to GenTeal® gel. Patients were prescribed GenTeal® gel and instructed to instill 1-2 drops of GenTeal® gel 2 to 4 times
daily for 20 weeks in the conjunctival cul-de-sac of both eyes. The dose selection was based on the ophthalmologist’s current clinical practice. No other tear substitute other than GenTeal® gel was allowed from enrollment until the end of study. The patients were assessed at three different visits at baseline (Day 0), (week 10-12) and at week 20.
The primary efficacy of the study was evaluated by assessing the change in dry eye symptoms calculated as individual symptom scores [(foreign body sensation, itching, burning, watering, photophobia and feeling of dryness in the eye); each on a scale 0-3; none (0), mild (1), moderate (2), severe (3)] and composite symptoms score which ranged from 0-18. In addition dry eye signs (i.e.; TBUT, Schirmer’s test, fluorescein corneal staining)19 were also performed at the discretion of the ophthalmologist from baseline and at week 20. The global assessment of treatment efficacy on dry eye conditions (i.e. Excellent, Good, Fair, Poor) was also assessed by the ophthalmologist as well as by the subjects.
The secondary objective of the study was to evaluate ocular tolerance and systemic safety of the product through global assessment of tolerability by the ophthalmologist & by the subjects: Ocular tolerance was assessed by enquiring about and recording ocular discomfort that occurred in relation to instillation of the study medication using a defined grading (e.g. excellent, good, fair, poor) and systemic safety was evaluated from AE reporting. Both the efficacy and safety variables were measured at all the visits.
| Table 1 Demographie summary of the patients | ||
|---|---|---|
| GenTeal® Gel treatment N=170 | ||
| Age (years) | Mean ± SD | 56.6± 9.42 |
| Median | 54.0 | |
| Range | 39.0-82.0 | |
| Age group – n (%) | 35-44 years | 11 (6.47%) |
| 45-54 years | 74 (43.5%) | |
| 55-64 years | 51 (30.0%) | |
| 75-85 years | 12 (7.06%) | |
| Other* | 3 (1.76%) | |
| Gender-n (%) | Female | 170 (100%) |
Analysis for efficacy and safety was performed for all evaluable patients. One-way ANOVA was applied for ocular signs (i.e. tear breakup time, Schirmer’s test score and Fluorescein staining) between various time intervals. Chisquare test was used for analysis of dry eye symptoms at various time intervals. All statistical tests were applied using SAS statistical software (version 9.) 2 and the p value of < 0.05 were considered statistically significant.
Of the 170 enrolled subjects, 169 completed the study and 1 subject discontinued from the study due to lost to follow up. The mean age of the 170 female subjects enrolled into the study was 56.6 ± 9.42 years with 12 subjects were more than 74 years of age. The demographic and other baseline details of signs and symptoms are given in Tables 1 & 2.
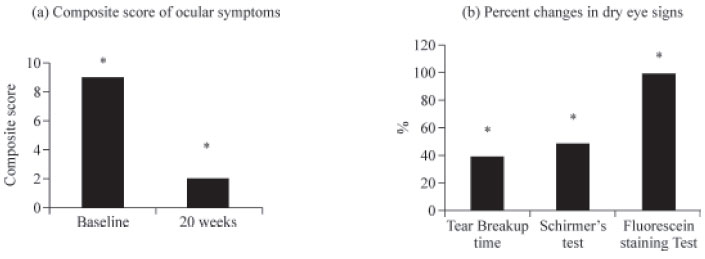
Treatment with GenTeal® gel by post menopausal women with dry eye syndrome for 20 weeks significantly reduced (p<0.0001) the composite score of dry eye symptoms (Table 3, Fig. 1a). All the symptoms of dry eye (i.e. burning sensation, foreign body sensation, itching, watering of eyes and dryness) except photophobia were significantly reduced at 20 weeks of treatment (Table 3). All the symptoms showed a decreasing pattern from baseline, at 10 weeks and at 20 weeks.
A significant improvement was observed in the dry eye signs with GenTeal® gel treatment over 20 weeks (Table 4). There was a significant increase in tear breakup time and Schirmer’s test score (p<0.0001) with significant decrease in Fluorescein staining cumulative score (p<0.0001) (Table 4, Fig. 1b). The overall efficacy with GenTeal® gel treatment for 20 weeks was ‘good’ to ‘excellent’ for >99% of the subjects as per both the physician’s and the subject’s assessment (Table 5).
| Table 2 Baseline disease characteristics of study subjects | ||||||
|---|---|---|---|---|---|---|
| Symptom Severity | ||||||
| None | Mild | Moderate | Severe | N | ||
| Symptoms- N(%) | Buming sensation | 11 (6.59%) | 45 (26.95%) | 85 (50.90%) | 26 (15.57%) | 167 |
| Foreign body sensation | 18 (11.04%) | 45 (27.61%) | 72 (44.17%) | 28 (17.18%) | 163 | |
| Itching | 17 (10.37%) | 45 (27.44%) | 67 (40.85%) | 35 (21.34%) | 164 | |
| Watering of eyes | 38 (23.75%) | 51 (31.88%) | 52 (32.50%) | 19 (11.88%) | 160 | |
| Dryness | 5 (3.18%) | 37 (23.57%) | 63 (40.13%) | 52 (33.12%) | 157 | |
| Photophobia | 68 (46.26%) | 47 (31.97%) | 19 (12.93%) | 13 (8.84%) | 147 | |
| Table 3 The change from baseline in the individual and composite scores of ocular symptoms with GenTeal® Gel treatment at 10 and 20 weeks | ||||||
|---|---|---|---|---|---|---|
| Burning sensation | Foreign body sensation | Itching | Watering of eyes | Dryness | Photophobia | |
| Baseline | ||||||
| Media (Range) (N) | 2 (0-3) (167) |
2 (0-3) (163) |
2 (0-3) (164) |
1 (0-3) (160) |
2 (0-3) (157) |
1 (0-3) (147) |
| Week 10: | ||||||
| Media (Range) | 1 (0-3) (169) |
1 (0-3) (164) |
1 (0-3) (165) |
1 (0-2) (161) |
1 (0-3) (161) |
0 (0-3) (150) |
| Media Change | 1 | 1 | 1 | 1 | 1 | 1 |
| Media % Change | 50 | 50 | 50 | 0 | 50 | 100 |
| Week 20 | ||||||
| Media (Range) | 0 (0-3) (168) |
0 (0-2) (163) |
0 (0-2) (164) |
0 (0-1) (159) |
0 (0-2) (161) |
0 (0-1) (150) |
| Media Change | 2 | 2 | 2 | 1 | 2 | 1 |
| Media % Change | 100 | 100 | 100 | 100 | 100 | 100 |
| P value | 0.000246 | 0.00681 | 0.00412 | 0.0356 | 0.00035 | 0.605 |
| Composite# score of the ocular symptoms | ||||||
| Median (Range) | Median % Change | |||||
| Baseline value (N=170) | 9 (2-18) | |||||
| At 10 weeks (N=170) | 4 (0-14) | 56 | ||||
| At 20 weeks (N=169) | 2 (0-9) | 78* | ||||
| *p<0.0001 as compared to baseline # The Composite score is calculated from the addition of all the six symptom scores and ranges from 0-18. |
||||||
None of the patient died or experienced serious adverse events (SAEs) during the study. Adverse events of dyspepsia and increased frequency of micturition were reported by a subject and they were of mild and moderate severity, respectively. The overall tolerability of GenTeal® gel, as per the assessment by both the patients and the physicians at 20 weeks was ‘good’ to ‘excellent’ for 98.8% and 100% of the subjects respectively (Table 6).
Dry eye conditions are the most common cause of red and irritated eyes in people over the age of 65 years20. As dry eye disease causes significant reduction in quality of life, limits daily activities, reduced social and physical functioning, and decreased workplace productivity21, 22. Among several risk factors for dry eye, age >50 years and female gender are considered important23, 24. Therefore current study was performed on post menopausal female subjects with a mean age of 56.6 ± 9.42 years.
In the present study; significant improvements were
The effect of GenTeal® Gel treatment on dry ocular score of dry eye symptoms and dry eye signs on 20 weeks.
a) Change in composite score of six dry eye symptoms (burning sensation, foreign body sensation, itching, watering of eyes and dryness and photophobia measure at a scale of 1-3),
b) Percent changes in dry eye signs performed by tear break up time, schirmer’s test and by fluorescein staining test
| Table 4 The change from baseline in the dry eye signs with GenTeal® Gel treatment at 10 and 20 weeks | |||
|---|---|---|---|
| Tear Breakup time (seconds) (Mean ± SD) (N) | Schirmer’s test (mm/5min) (Mean ± SD) (N) | Fluorescein staining cumulative score # Median (Range) (N) | |
| Baseline: | 7.22±2.40 (107) | 6.80±3.64 (132) | 4.00(0.00-9.00) (63) |
| Week10: | 8.92 ± 2.63 (101) | 9.06 ± 4.11 (126) | 2.00(0.00-9.00) (61) |
| % change | -23.5 | -33.2 | 50.0 |
| Week20: | 10.1.92 ± 2.77 (95) | 10.7 ± 2.77 (120) | 0.00(0.00-0.00) (60) |
| % change | -39.9 | -48.6 | 100.0 |
| p value | <0.0001 | <0.0001 | <0.0001 |
| *p<0.0001 as compared to baseline # The Cumulative score (0 to 9 is calculated by adding the staining score of all three zones (0 to 3) | |||
reported in incidence of foreign body sensation itching, burning, watering and feelings of dryness with GenTeal® gel treatment. However a non significant improvement was observed for photophobia, one of the dry eye symptoms. Results of the current study are in consistent with an earlier study where GenTeal® gel treatment showed approximately 33 % reduction at 4 weeks of treatment in symptoms score17. However the greater reduction (78%) in symptom scores might be due to longer duration of treatment for 20 weeks in the present study. Likewise, the significant improvement in mean TBUT (39.9%) in our study is nearly similar to those found in earlier study with GenTeal® gel
| Table 5 Global assessment of overall efficacy at 20 weeks – n (%) (ITT population) | |
|---|---|
| Overall efficacy | GenTeal® Gel |
| Number of patient’s n (%) | N=169 |
| Patient’s assessment | |
| Excellent | 44 (26.19%) |
| Very good | 70 (41.67%) |
| Good | 53 (31.55%) |
| Poor | 1 (0.60%) |
| Physician’s assessment | |
| Excellent | 49 (29.17%) |
| Very good | 75 (44.64%) |
| Good | 44 (26.19%) |
| Poor | 0 (0.00%) |
| Table 6 Global assessment of overall tolerability at 20 weeks | |
|---|---|
| Overall tolerability | GenTeal® Gel |
| Number of patient’s n (%) | N=169 |
| Patient’s assessment | |
| Excellent | 52 (30.77%) |
| Very good | 69 (40.83%) |
| Good | 46 (27.22%) |
| Poor | 2 (1.18%) |
| Physician’s assessment | |
| Excellent | 61 (36.09%) |
| Very good | 62 (36.69%) |
| Good | 46 (27.22%) |
| Poor | 0 (0.00%) |
(59%) in US patients after 4 weeks treatment. Importantly, our study included much larger number of patients (n = 169) in the effectiveness analyses compared with the above clinical practice study (n = 37)17. The fact that TBUT improvement was accompanied by subjective symptom improvement suggests that the change in TBUT was clinically meaningful.
The improvements in Schirmer scores and fluorescein staining score of treatment in our study are in agreement with those determined in the earlier trial of GenTeal® gel17.
Additionally the efficacy of GenTeal gel was also supported by the global evaluation of efficacy as rated ‘good’ to ‘excellent’ for >99% of the subjects as per both the physician’s and the subject’s assessment. These results are also similar with the findings of a previous study, in which global evaluation of efficacy was much better for the 78% of patients at 4 weeks after treatment initiation17. Improvement in ocular symptoms and signs of dry eye suggests that GenTeal® gel treatment.
Therefore, we speculate that GenTeal® gel partially resolves the pathophysiological changes causing dry eye disease and prevents further damage to the ocular surface thereby drastically improve the quality of life of elderly individuals.
Study observed that GenTeal® gel treatment was welltolerated with only positive experiences by most of the patients. The adverse events of dyspepsia and frequent micturition were reported only by one in patient. No patient discontinued treatment because of adverse events in our study. These are in tune with results of a clinical practice studies performed in the United States17.
Study has few limitations of being non-comparative and of open-label design; however despite these limitations, the use of multiple objective tests (i.e., TBUT, Schirmer and conjunctival staining scores) and subjective measures (i.e., ocular symptom scores) has enabled us to provide clinically relevant evidence of the effectiveness of GenTeal® gel for treatment of dry eye disease. However, the clinical practice setting enhances the external validity of our findings, as does the prospective, comparator arm, surveillance design.
In conclusion, the effectiveness and well tolerability findings of our study support the use of GenTeal® gel for the treatment of moderate to severe dry eye disease in routine clinical practice setting.
*GenTeal Study Group : Dr Niteen C. Dedhia, Dr Kumar Doctor, Dr Himanshu Mehta, Dr Quresh Maskati, Dr Hijab Mehta, Dr Pawan Goyal, Dr Lopmundra Das, Dr A. K. Bakshi, Dr Saurabh Chaudhry, Dr Rakesh Mahajan, Dr Rajneesh Chadha, Dr Vikas Mittal, Dr Kummararaj G., Dr V Basten, Dr Rekha, Dr Souvic Banerjee, Dr Ranjan Kumar Akhauri, Dr N Borah.